
Food preparation, for example, offers an appropriate comparison. The general approach to using stoichiometric relationships is similar in concept to the way people go about many common activities. In this module, the use of balanced chemical equations for various stoichiometric applications is explored. These quantitative relationships are known as the reaction’s stoichiometry, a term derived from the Greek words stoicheion (meaning “element”) and metron (meaning “measure”). Coefficients provide the relative numbers of these chemical species, allowing a quantitative assessment of the relationships between the amounts of substances consumed and produced by the reaction. Chemical formulas provide the identities of the reactants and products involved in the chemical change, allowing classification of the reaction. Perform stoichiometric calculations involving mass, moles, and solution molarityĪ balanced chemical equation provides a great deal of information in a very succinct format.Use balanced chemical equations to derive stoichiometric factors relating amounts of reactants and products.  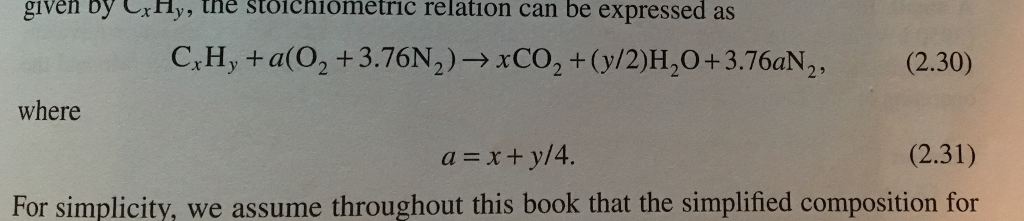
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
